Abstract
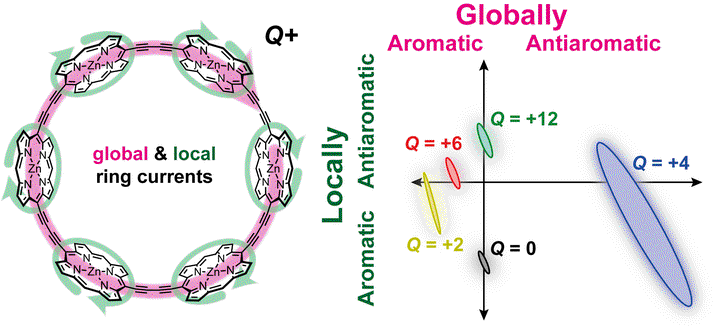
Magnetic field-induced ring currents in aromatic and antiaromatic molecules cause characteristic shielding and deshielding effects in the molecules’ NMR spectra. However, it is difficult to analyze (anti)aromaticity directly from experimental NMR data if a molecule has multiple ring current pathways. Here we present a method for using the Biot-Savart law to deconvolute the contributions of different ring currents to the experimental NMR spectra of polycyclic compounds. This method accurately quantifies local and global ring current susceptibilities in porphyrin nanorings, as well as in a bicyclic dithienothiophene-bridged [34]octaphyrin. There is excellent agreement between ring current susceptibilities derived from both experimental and computationally-predicted chemical shifts, and with ring currents calculated by the GIMIC method. Our method can be applied to any polycyclic system, with any number of ring currents, provided that appropriate NMR data are available.